Sterility testing plays a critical role in pharmaceutical quality control. The test confirms that sterile products do not contain viable microorganisms that could harm patients. Pharmacopeias such as USP, EP, and IP provide harmonized guidelines that manufacturers must follow when performing sterility tests.
Most microbiologists know that sterility testing normally requires two culture media. One medium supports bacterial growth, while the other supports fungi and aerobic organisms. However, a special situation arises when a product contains mercurial preservatives. These preservatives can interfere with microbial recovery and create challenges during sterility testing.
Pharmacopeias recognize this issue and provide an alternative approach. Under certain conditions, laboratories may use Fluid Thioglycollate Medium (FTM) instead of Soybean-Casein Digest Medium (SCDM). Understanding this exception helps microbiologists design compliant and scientifically sound sterility tests.
Standard Media Used in Sterility Testing
Pharmacopeial sterility testing requires two different media to ensure the detection of a wide range of microorganisms.
Fluid Thioglycollate Medium (FTM) supports the growth of aerobic and anaerobic bacteria. Laboratories usually incubate this medium at 30–35°C.
Soybean-Casein Digest Medium (SCDM) supports the growth of fungi and aerobic bacteria. Analysts normally incubate this medium at 20–25°C.
This approach increases the probability of detecting bacterial and fungal contamination. Most pharmaceutical sterility tests follow this standard setup.
The Problem with Mercurial Preservatives
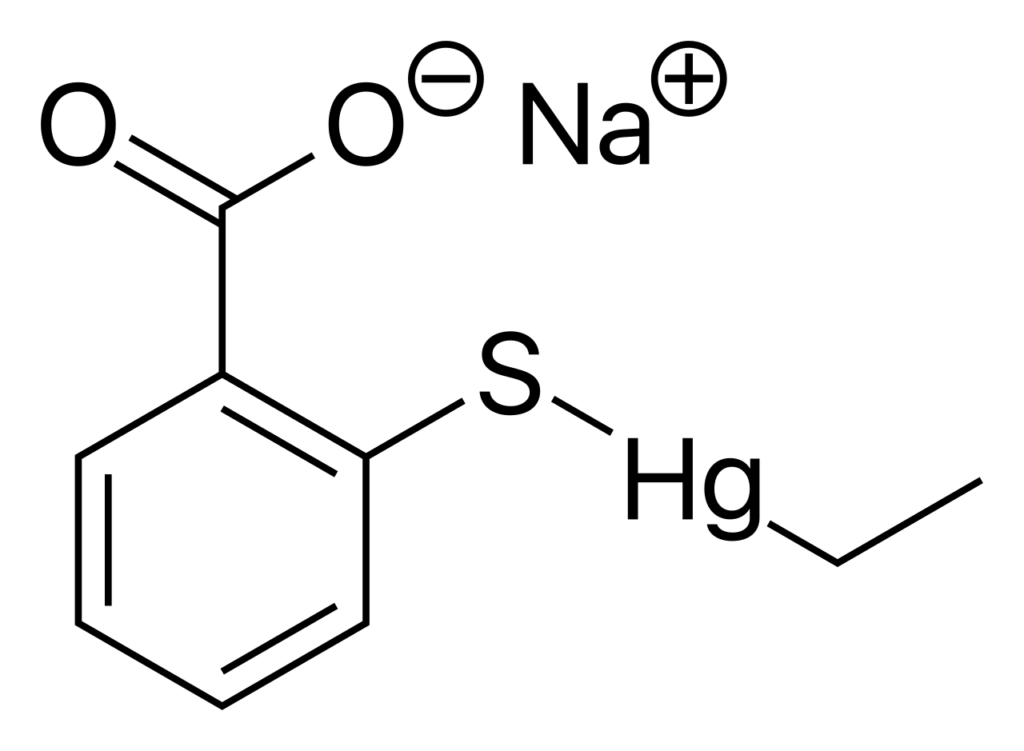
Some pharmaceutical formulations contain mercurial preservatives such as thiomersal (thimerosal). Manufacturers add these compounds to prevent microbial contamination during storage and use.
Mercurial preservatives possess strong antimicrobial activity. They can suppress or completely inhibit microbial growth in sterility test media. This inhibition creates a serious challenge during sterility testing.
When analysts introduce a product containing mercurial preservatives directly into Soybean-Casein Digest Medium, the preservative may inhibit fungal growth. Even if fungi exist in the sample, the preservative may prevent their recovery. This situation can produce a false negative sterility result.
Such interference directly affects the reliability of the sterility test.
Pharmacopeial Guidance for Mercurial Preservatives
Pharmacopeias address this challenge in the harmonized sterility testing chapters such as USP <71> Sterility Tests, the European Pharmacopoeia section on sterility testing, and the Indian Pharmacopoeia sterility testing chapter.
These guidelines recognize that mercurial preservatives can inhibit microbial recovery in certain media. Therefore, they allow laboratories to modify the test method when justified.
If membrane filtration cannot remove the preservative effectively, the pharmacopeia allows laboratories to replace Soybean-Casein Digest Medium with Fluid Thioglycollate Medium incubated at a lower temperature.
In this scenario, the laboratory incubates FTM at 20–25°C instead of using SCDM.
This adjustment allows the medium to support the recovery of fungi and aerobic microorganisms while avoiding the inhibitory interaction that occurs in SCDM.
Why Fluid Thioglycollate Medium Works
Fluid Thioglycollate Medium contains reducing agents that create a favorable environment for microbial recovery. The composition of this medium often tolerates inhibitory substances better than SCDM.
The medium contains sodium thioglycollate and L-cystine, which act as strong reducing agents. These compounds contain sulfhydryl (–SH) groups that react with mercury ions and convert them into less toxic complexes. This reaction lowers the antimicrobial activity of mercurial preservatives such as thiomersal (thimerosal), allowing microorganisms to survive and grow in the medium.
When analysts incubate FTM at 20–25°C, the medium can support fungal growth along with aerobic microorganisms. This incubation condition effectively replaces the role of SCDM in the sterility test.
This approach does not weaken the sterility test. Instead, it maintains microbial detection capability when the product formulation interferes with standard media.
Validation Requirement for Media Substitution
Pharmacopeias do not allow laboratories to change sterility test media without scientific justification. The laboratory must validate the modified method before routine use.
Method suitability testing confirms that the medium can recover specific microorganisms in the presence of the product.
During validation, analysts must demonstrate successful recovery of the following challenge organisms:
- Staphylococcus aureus
- Pseudomonas aeruginosa
- Clostridium sporogenes
- Bacillus subtilis
- Candida albicans
- Aspergillus brasiliensis
Inoculate small numbers of these organisms into the sterility test system containing the product. The organisms must grow successfully in the chosen medium. Successful recovery proves that the preservative does not inhibit microbial detection under the validated conditions.
Once the laboratory completes this validation, it can use the modified sterility test method during routine analysis.