Pharmaceutical products are expected to be safe for patients. However, not all medicines need to be sterile. Many dosage forms such as tablets, capsules, syrups, ointments, and creams are manufactured as non-sterile products.
Even though they do not require sterility, manufacturers must still control microbial contamination. Excessive microbial growth or the presence of pathogenic microorganisms can make the product unsafe and unstable.
To ensure safety, pharmaceutical companies perform microbiological tests for non-sterile products, commonly known as Microbial Limit Tests (MLT). These tests determine how many microorganisms are present in the product and whether harmful microorganisms are absent.
Let’s explore the microbiological tests for non-sterile pharmaceutical products, the regulatory requirements, and how to perform them in a pharmaceutical microbiology laboratory.
Why To Perform MLT Required for Non-Sterile Products
Non-sterile pharmaceutical products are exposed to several potential sources of contamination during manufacturing. Raw materials, water systems, equipment, packaging materials, and even the manufacturing environment can introduce microorganisms.
If microbial contamination is not controlled, it may cause several problems:
Product spoilage
Reduced shelf life
Formation of toxins
Patient infections
Product recalls
Regulatory agencies, therefore, require pharmaceutical manufacturers to control the microbiological quality of non-sterile products.
The concept behind microbial testing is simple. Non-sterile products may contain small numbers of microorganisms, but the levels must remain within acceptable limits and dangerous pathogens must not be present.
These requirements are defined in pharmacopeial guidelines such as:
- USP microbiology chapters
- European Pharmacopoeia
- Japanese Pharmacopoeia
The commonly referenced USP chapters include USP <61>, USP <62>, and USP <1111>.
What Are Microbial Limit Tests?
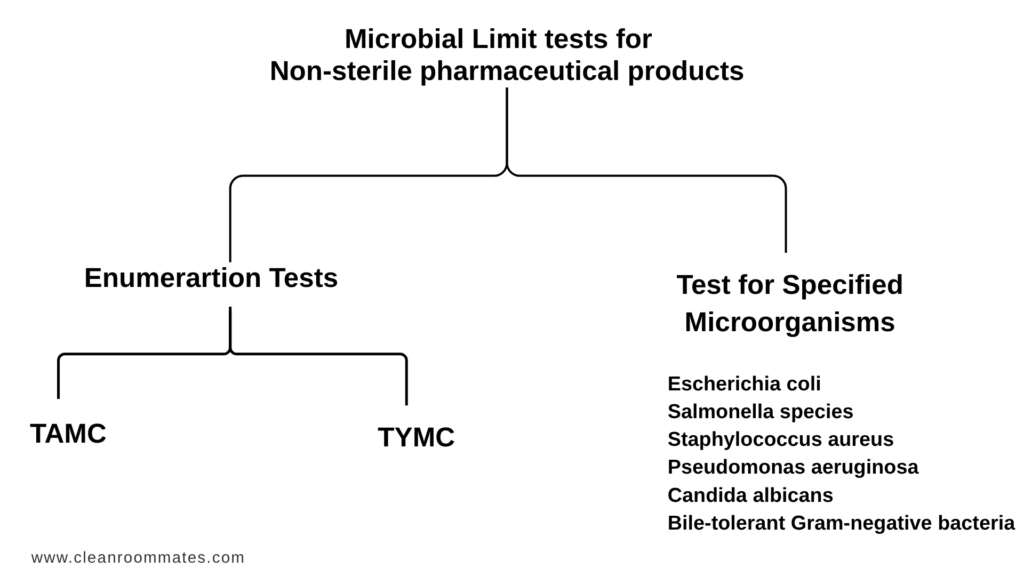
Microbial Limit Tests consist of two main parts.
First, the determination of the total number of microorganisms present in the product.
Second, they check whether specific harmful microorganisms are present.
Therefore, microbial testing for non-sterile pharmaceutical products generally includes:
- Microbial enumeration tests
- Tests for specified microorganisms
These tests ensure that microbial contamination remains within safe limits.
Microbial Enumeration Tests (USP <61>)
Microbial enumeration tests determine the total microbial load present in the product. These tests measure the number of viable microorganisms in a sample and report the result as colony forming units (CFU).
The enumeration test mainly measures two parameters.
Total Aerobic Microbial Count (TAMC)
This test measures the number of aerobic bacteria present in the product.
Total Yeast and Mold Count (TYMC)
This test measures the number of fungal organisms such as yeasts and molds.
Both values provide an overall idea of microbial contamination in the product.
Methods Used for Enumeration
Several methods are used to determine microbial counts.
The most common methods include:
Membrane filtration
Pour plate method
Spread plate method
The method used depends on the nature of the product. Liquid products are often tested using membrane filtration, while powders and creams may be tested using plating methods.
Sample Preparation
Before performing enumeration tests, the product sample is diluted using sterile diluents. Dilution is important because high microbial counts can make colonies difficult to count.
Sometimes the product contains antimicrobial preservatives. In such cases, add neutralizers to either the media or the diluents to prevent inhibition of microbial growth.
Tests for Specified Microorganisms (USP <62>)
Enumeration tests tell us how many microorganisms are present. However, some microorganisms are particularly dangerous even in very small numbers.
For this reason, pharmacopeias require testing for specified microorganisms, also called objectionable organisms.
These tests determine whether certain pathogens are present in the product.
Common microorganisms tested include:
Escherichia coli
Salmonella species
Staphylococcus aureus
Pseudomonas aeruginosa
Candida albicans
Bile-tolerant Gram-negative bacteria
The presence of these organisms indicates poor hygiene, contamination, or inadequate manufacturing controls.
General Procedure
The test usually begins with an enrichment step. Inoculate the sample into a suitable broth that promotes microbial growth.
After incubation, streak the culture onto selective agar media. These media help detect specific organisms based on colony appearance and biochemical reactions.
If no colonies appear or the colonies do not match the characters, the sample passes the test.
Acceptance Criteria for Non-Sterile Products
The acceptable microbial limits depend on the type of product and its route of administration.
Products that are consumed orally generally allow higher microbial limits compared to products applied to sensitive areas such as the eyes or nasal cavity.
For example, oral solid dosage forms often allow:
- TAMC up to 10³ CFU per gram
- TYMC up to 10² CFU per gram
- Specified microorganisms must be absent
These limits are defined in pharmacopeial guidelines such as USP <1111> Microbiological Quality of Non-Sterile Pharmaceutical Products.
Manufacturers must define microbial specifications based on these guidelines and include them in product quality standards.
Method Suitability Testing
Method suitability is a critical step before performing microbial limit tests.
Some pharmaceutical products contain preservatives, antimicrobial agents, or high sugar concentrations. These substances can inhibit microbial growth during testing.
If microorganisms fail to grow during the test, it may produce false negative results.
To prevent this issue, laboratories perform method suitability tests. Inoculate the product with known microorganisms to confirm that the test method can recover them successfully.
If recovery is poor, modify the test method by adjusting dilution, filtration, or neutralization steps.
Importance of Microbial Identification
When microbial colonies grow during testing, microbiologists often identify them.
Identification helps in several ways.
It helps determine whether the organism is objectionable.
It helps trace the contamination source.
It helps improve contamination control strategies.
Modern laboratories use technologies such as MALDI-TOF, VITEK, or molecular identification methods for accurate microbial identification.
Common Challenges in Microbial Limit Testing
Microbiological testing for non-sterile products can be challenging for several reasons.
Products with strong preservatives may inhibit microbial growth during testing.
Highly viscous products such as ointments and creams are difficult to dilute and filter.
Some microorganisms grow slowly and may be missed during incubation.
For this reason, laboratories must carefully validate their testing methods and follow microbiological procedures correctly.
Regulatory Expectations
Regulatory authorities expect pharmaceutical companies to maintain strict microbiological control of non-sterile products.
Regulatory auditors usually review:
Microbial specifications
Test methods
Method suitability data
Environmental monitoring results
Investigation reports for microbial failures
Frequently Asked Questions (FAQs)
Microbiological tests for non-sterile pharmaceutical products are laboratory tests used to determine the microbial quality of medicines that are not required to be sterile. These tests measure the total number of microorganisms present and confirm the absence of harmful pathogens that may pose risks to patients.
A microbial limit test is a microbiological examination performed on non-sterile pharmaceutical products to determine the acceptable number of microorganisms present in the product. It includes microbial enumeration tests and tests for specified microorganisms to ensure the product meets pharmacopeial microbial quality standards.
TAMC stands for Total Aerobic Microbial Count, which measures the total number of aerobic bacteria in a pharmaceutical product. TYMC stands for Total Yeast and Mold Count, which measures fungal contamination such as yeasts and molds. Both parameters are used to evaluate microbial contamination levels in non-sterile products.
Microbiological testing of non-sterile pharmaceutical products is described in the following pharmacopeial chapters:
USP <61> Microbiological Examination of Nonsterile Products – Microbial Enumeration Tests
USP <62> Microbiological Examination of Nonsterile Products – Tests for Specified Microorganisms
USP <1111> Microbiological Quality of Nonsterile Pharmaceutical Products
These chapters define testing methods and acceptable microbial limits.
Pharmaceutical microbiology laboratories test for several specified microorganisms that may pose health risks. These commonly include Escherichia coli, Salmonella, Staphylococcus aureus, Pseudomonas aeruginosa, and Candida albicans depending on the product type.
Method suitability ensures that the test method can detect microorganisms in the presence of the product. Some pharmaceutical products contain preservatives or antimicrobial ingredients that inhibit microbial growth. Method suitability testing confirms that microorganisms can still be recovered during laboratory testing.
Acceptable microbial limits vary depending on the product type and route of administration. For example, many oral solid dosage forms typically allow up to 10³ CFU/g for TAMC and 10² CFU/g for TYMC, while products used in sensitive areas such as nasal or ophthalmic applications have stricter limits.
Sterile pharmaceutical products must be completely free from all microorganisms, while non-sterile products are allowed to contain small numbers of microorganisms within acceptable limits. However, non-sterile products must not contain harmful or objectionable microorganisms that could affect patient safety.
Yes, microbial identification is often performed when colonies grow during testing. Identification helps determine whether the microorganism is objectionable and helps microbiologists trace the contamination source within the manufacturing process.



