In the pharmaceutical industry, we do a lot of documentation in our day-to-day work, from entry logs to batch manufacturing records. Good Documentation Practices (GDP) tell us that every activity must be recorded at the time it occurs, which is called contemporaneous recording under the ALCOA principles (Attributable, Legible, Contemporaneous, Original, and Accurate) in pharma documentation.
In fact, it is said that if it’s not documented, it’s not done. That’s how critical documentation is in pharma.
As human beings, we certainly make mistakes. That is exactly why companies develop clear procedures to correct errors in documentation. At the same time, ensuring data integrity is also essential.
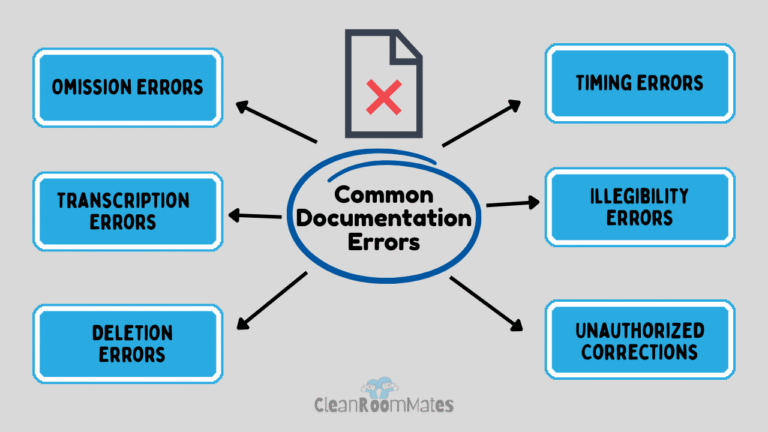
These documentation procedures define what types of entry errors can happen (like omissions, calculation mistakes, or transcription errors) and how to correct them transparently.
Considering the importance of documentation, each operator will undergo training on these procedures to follow when they make an error. However, it’s important to understand that these procedures are not meant to encourage all types of errors or enable data manipulation.
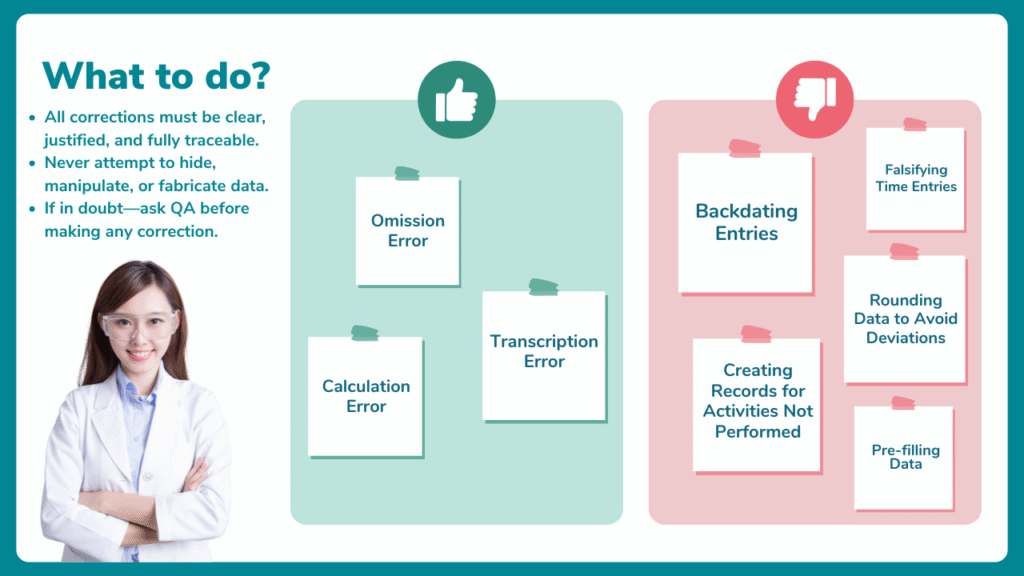
What is NOT Acceptable?
In a sterile manufacturing facility, the entry procedure describes the gowning steps, which take approximately 10 minutes if performed correctly.
If an operator enters the facility at 10:30 and records that time in the entry logbook, but completes gowning in only 2 minutes, this is a deviation from the procedure. He then enters the cleanroom and begins working on the equipment. He records the equipment logbook entry at 10:35. This indicates it took only 5 minutes from entry to starting work.
Under normal conditions, this process should take more than 10 minutes. This suggests that the gowning procedure was not correctly followed.
Realizing this discrepancy later, the operator corrects the equipment logbook entry time to 10:42 to create the appearance of compliance. Of course, he follows the documentation procedure to make this correction.
However, this is not an acceptable error or correction. The correction in time is an example of data falsification.
Let’s see how?
Whenever an operator enters time, it must be recorded accurately and contemporaneously, looking at the clock at that moment. There is no chance of recording false time unless the clock shows a false time. A deviation will be logged to justify the time correction in this case.
Each entry error must be strongly justified. If an error occurs, a supervisor or auditor should be able to easily see the circumstances and decide whether it was an honest mistake or an intentional act.
Even though standard operating procedures describe how to correct entry errors, it does not mean operators are free to make errors and simply correct them.
What Kind of Entry Errors Are Acceptable?
While errors should always be minimized, certain unintentional, honest mistakes are considered acceptable if they are corrected transparently and adequately documented.
Here are a few examples:
Transcription Errors
While copying results from an instrument printout to a logbook, an analyst record “21.8°C” instead of “22.8°C.”
The analyst notices the discrepancy during a routine check, draws a single line through the wrong entry, writes the correct value next to it, initials and dates the correction, and references the printout as justification.
This is acceptable because the error was unintentional, detected, and corrected transparently.
Omission Errors
An operator completes equipment cleaning but forgets to enter the cleaning start time. During a shift handover check, the supervisor notices the missing time.
The operator writes a comment stating, “Cleaning start time accidentally omitted. Actual start time was 14:00 per cleaning schedule,” signs and dates the note, and gets supervisor verification. The supervisor can acknowledge the correction by verifying the cleanliness of the equipment and the following actions performed on the equipment.
If the error is identified at a later date, it should be corrected at the current date.
This is acceptable as an omission error with documented justification.
Calculation Errors
A technician calculates the quantity of a buffer solution and records “12.0 L” instead of the correct “10.0 L.” The error is identified during a review.
A correction is made using the proper GDP procedure (single-line cross-out, correct value, initials, date, and reference to calculation).
This is acceptable because the error was not deliberate, and a clear audit trail exists.
In all these cases, the key principles that make the error acceptable are:
- The error was unintentional.
- It was identified and corrected as soon as possible.
- The original entry remains legible.
- The reason for the correction is documented.
- Corrections are signed and dated.
Conclusion
Entry errors in pharma documentation are sometimes unavoidable because we are human. However, only unintentional, properly justified, and transparently corrected errors are acceptable.
Any attempt to manipulate data, falsify records, or cover up deviations is strictly prohibited and can lead to regulatory action.
Remember:
- Always record data at the time of activity.
- Always check your entries carefully.
- Always follow GDP when correcting errors.
- Never assume any error can simply be “fixed later.”
- Follow the documentation procedures correctly. When in doubt, report to your supervisor immediately.
If you consistently apply these principles, your documentation will remain credible, compliant, and trustworthy.