When people think about cleanrooms, they often imagine sterile walls, HEPA filters, and strict gowning procedures. But one invisible factor quietly controls contamination every second—differential pressure.
If you work in pharmaceutical manufacturing, you already know the importance of contamination control. Differential pressure is not just a parameter that is displayed on a screen. It actively protects your process, your product, and your compliance status.
Let’s see how?

What is Differential Pressure in a Cleanroom?
Differential pressure refers to the pressure difference between two adjacent areas. In a cleanroom, this difference ensures that air always moves from the clean area to the dirt area. In other words, from higher cleanliness (higher pressure) to lower cleanliness (lower pressure).
Instead of allowing uncontrolled airflow, cleanrooms create a directional movement of air. This controlled airflow prevents contaminants from entering critical zones.
For example, a Grade B area maintains higher pressure than a Grade C area. Air flows outward when doors open, which blocks contaminated air from entering the cleaner space.
Why Differential Pressure Matters in Pharma
Pharmaceutical manufacturing depends heavily on environmental control. Even a small contamination event can lead to batch rejection, regulatory observations, or product recalls.
Differential pressure serves as one of the barriers controlling contamination. It does not physically block contaminants, but it controls airflow in a way that keeps contaminants away from critical zones.
When pressure differences drop or reverse, the system fails. Contaminated air can enter high-grade areas, increasing microbial load and particulate contamination.
This is why regulatory bodies expect continuous monitoring and strict control of pressure differentials in cleanrooms.
How Differential Pressure Controls Contamination
We all know that air always moves from high-pressure areas to low-pressure areas. Cleanroom design uses this principle to maintain cleanliness levels.
In a properly designed system, cleaner rooms maintain higher pressure compared to adjacent less clean rooms. This setup ensures that when a door opens, air rushes outward instead of inward.
This outward airflow prevents particles, microbes, and other contaminants from entering the critical area.
Without proper pressure differentials, even the best filtration system cannot fully protect the environment.
Pressure Cascading
Cleanrooms follow a cascading pressure pattern. This means pressure gradually decreases from the most critical area to the least critical area.
For example, a sterile filling area maintains the highest pressure. Surrounding rooms such as preparation and corridors maintain slightly lower pressure.
This stepwise reduction creates a protective airflow path. It ensures that contamination cannot travel backward into critical zones.
Pressure cascading forms the backbone of contamination control strategy in pharmaceutical facilities.
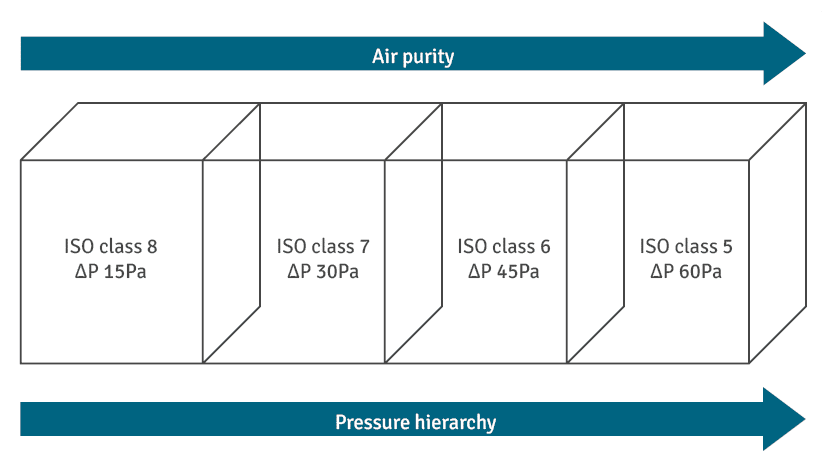
Recommended Pressure Differentials
Most pharmaceutical guidelines recommend a pressure difference of around 10 to 15 Pascals between adjacent areas.
For example, if a grade B area has a pressure differential of 45 Pa, the adjacent grade C should have around 30 Pa.
This range ensures adequate airflow without causing turbulence or operational difficulties like door opening issues.
However, each facility must justify its pressure limits based on design, process requirements, and risk assessment.
You should always validate your pressure differentials during qualification stages and verify them during routine monitoring as part of environmental monitoring.
Monitoring and Control of Differential Pressure
Modern cleanrooms use digital manometers, magnehelic gauges, or building management systems to monitor pressure differences.
Operators should regularly check readings and record them as part of environmental monitoring.
Alarms play a crucial role here. When pressure falls below or rises above limits, the system should alert personnel immediately.
Quick response prevents contamination risks and ensures compliance.
Common Issues and Their Impact
Pressure imbalance can occur due to several reasons. Door openings, HVAC failures, clogged filters, or improper air balancing can disrupt the system.
When differential pressure drops, the cleanroom loses one of its protective barriers. Contaminated air can enter, which increases microbial counts and particle levels.
Frequent fluctuations indicate deeper problems in HVAC design or maintenance practices.
Ignoring these issues can lead to audit observations and product quality risks.
Best Practices for Maintaining Differential Pressure
A well-designed HVAC system forms the foundation of pressure control. Regular maintenance ensures that filters, dampers, and fans function properly.
Operators should minimize unnecessary door openings. Airlocks should function correctly to maintain pressure integrity.
Operators shall not enter the cleanroom when the differential pressure is outside the established limits, and an alarm condition is active.
Routine calibration of pressure gauges ensures accurate readings. Trending data helps identify gradual changes before they become critical issues.
A proactive approach always works better than reactive corrections.
Without proper pressure differentials, even advanced cleanroom designs cannot maintain required cleanliness levels.
What EU GMP Annex 1 Says About Pharmaceutical Quality System
Disinfectants in Pharmaceutical Cleanrooms



